Some slides made using Biorender.com.
Skip to content
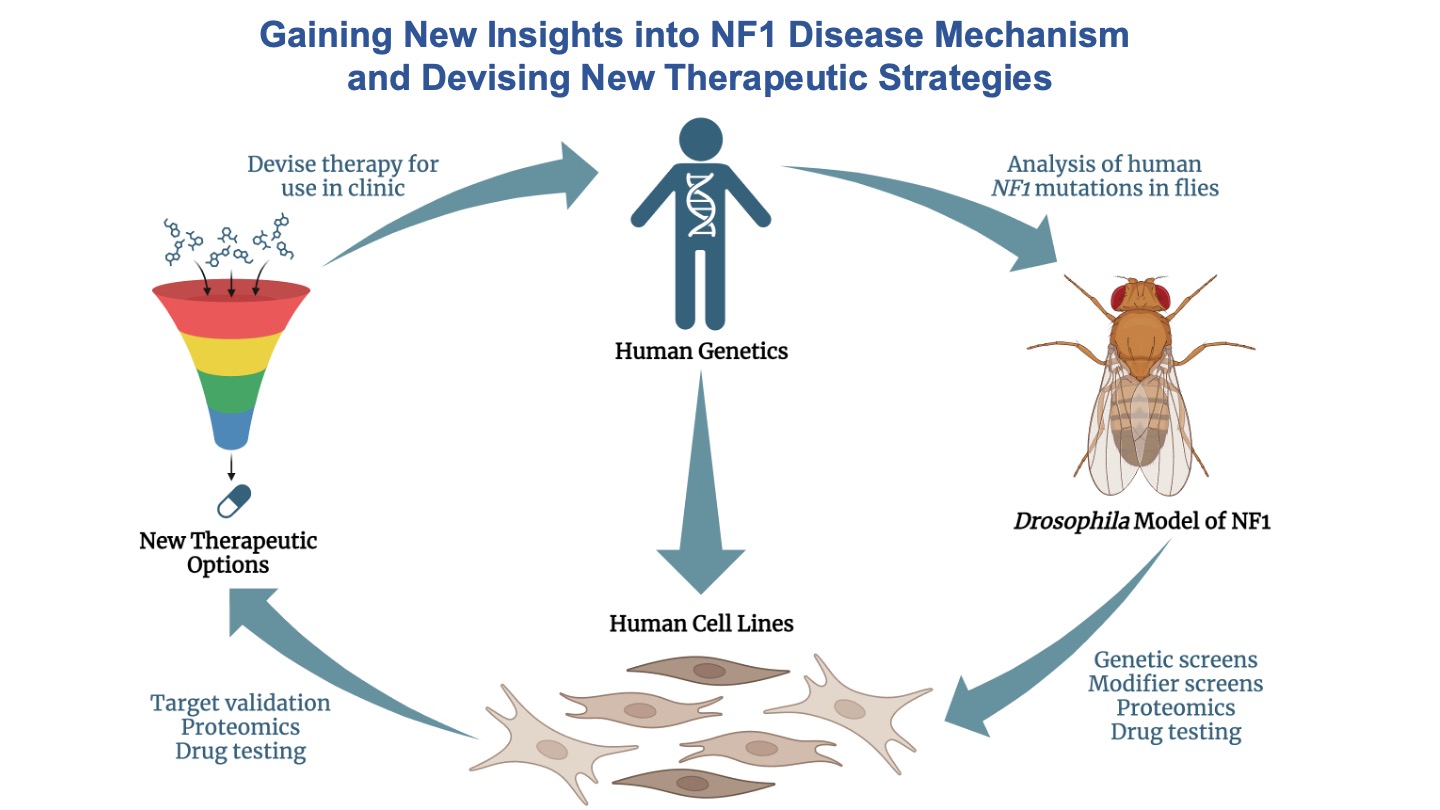
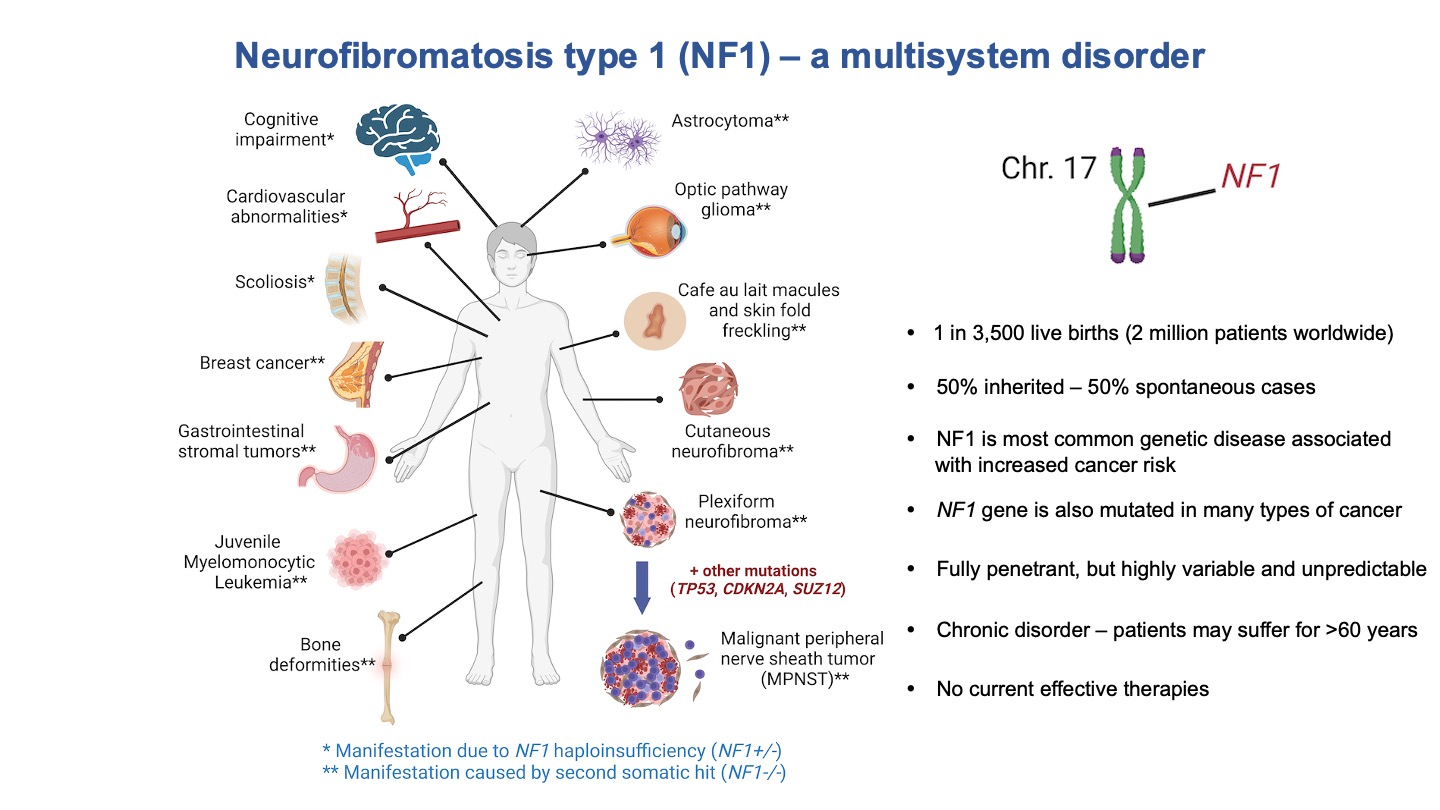
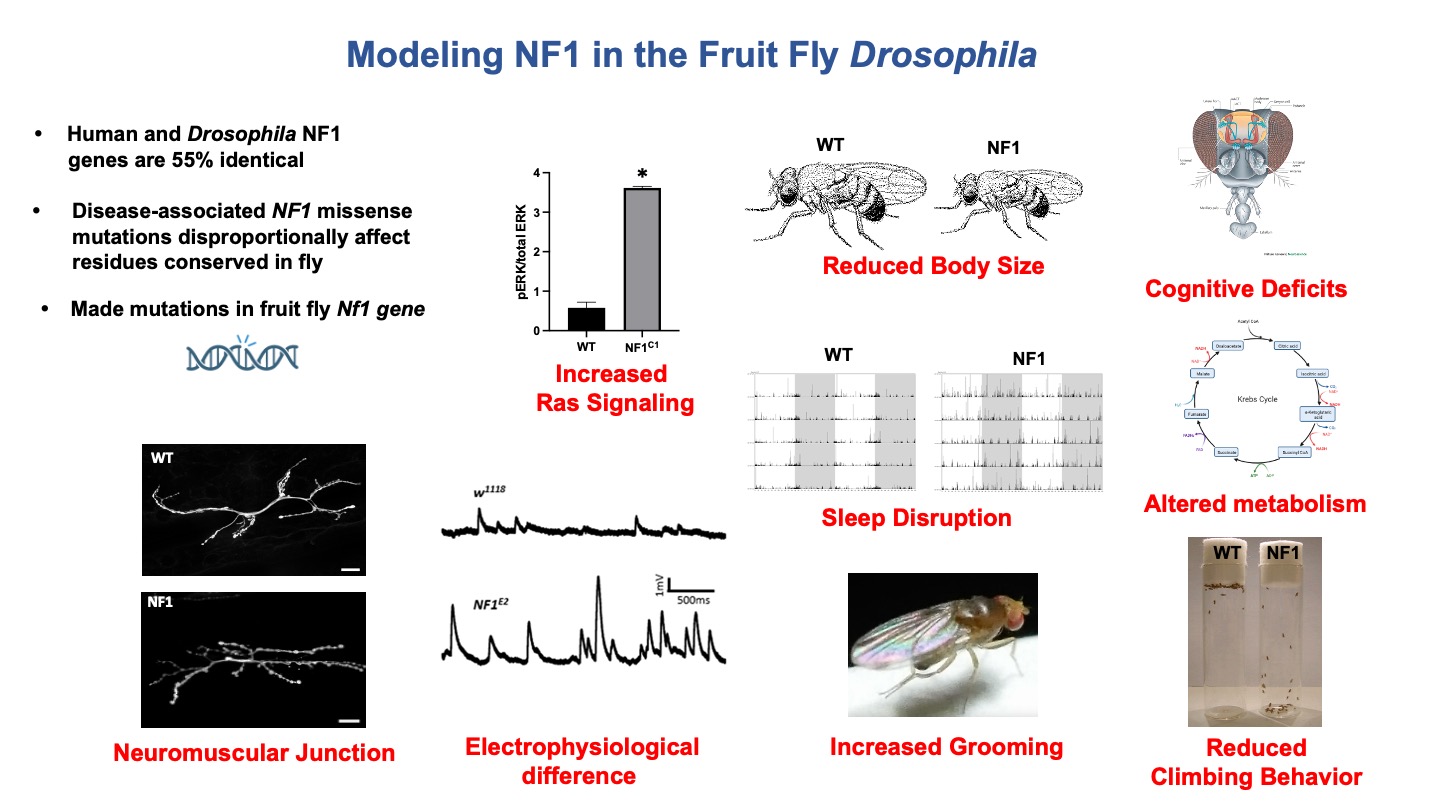

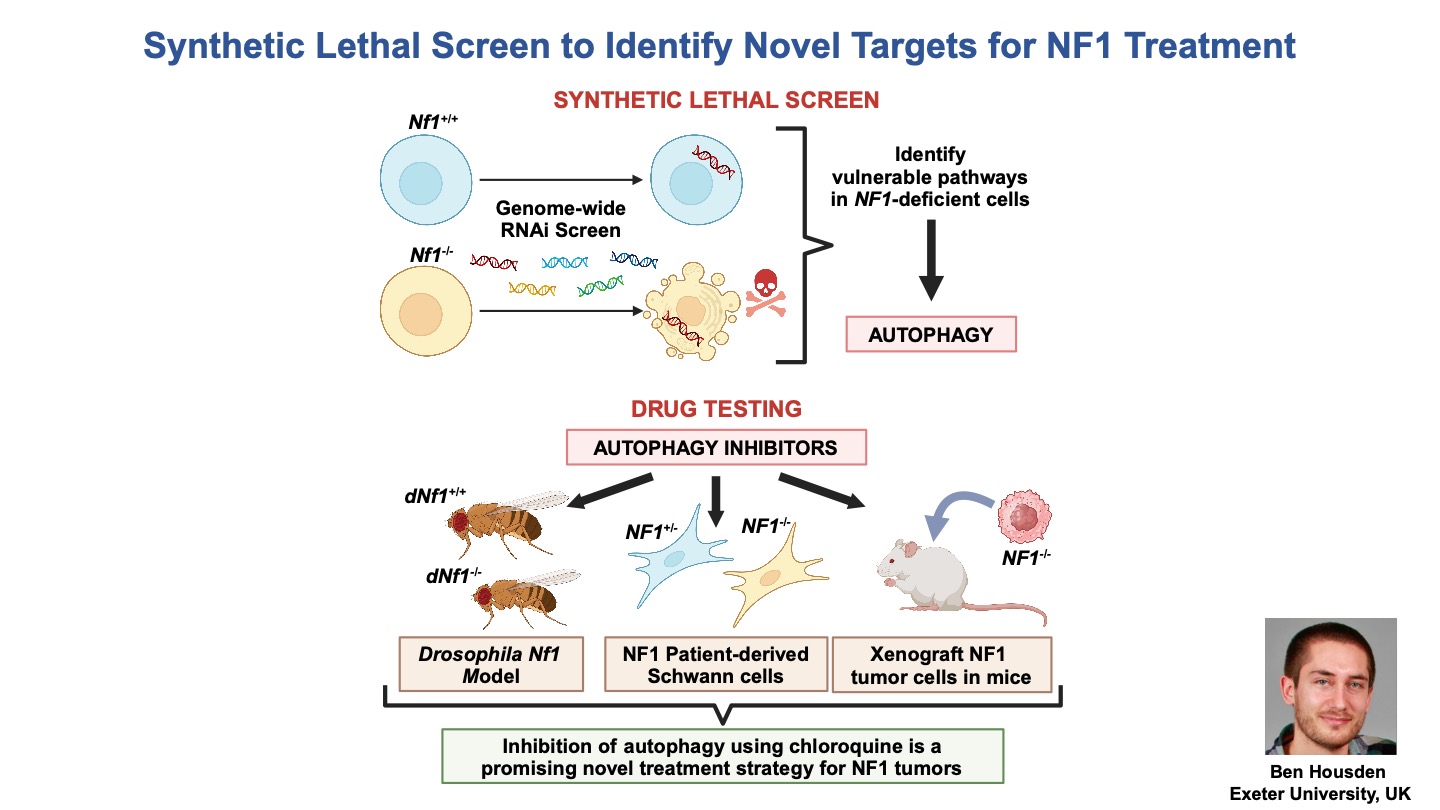
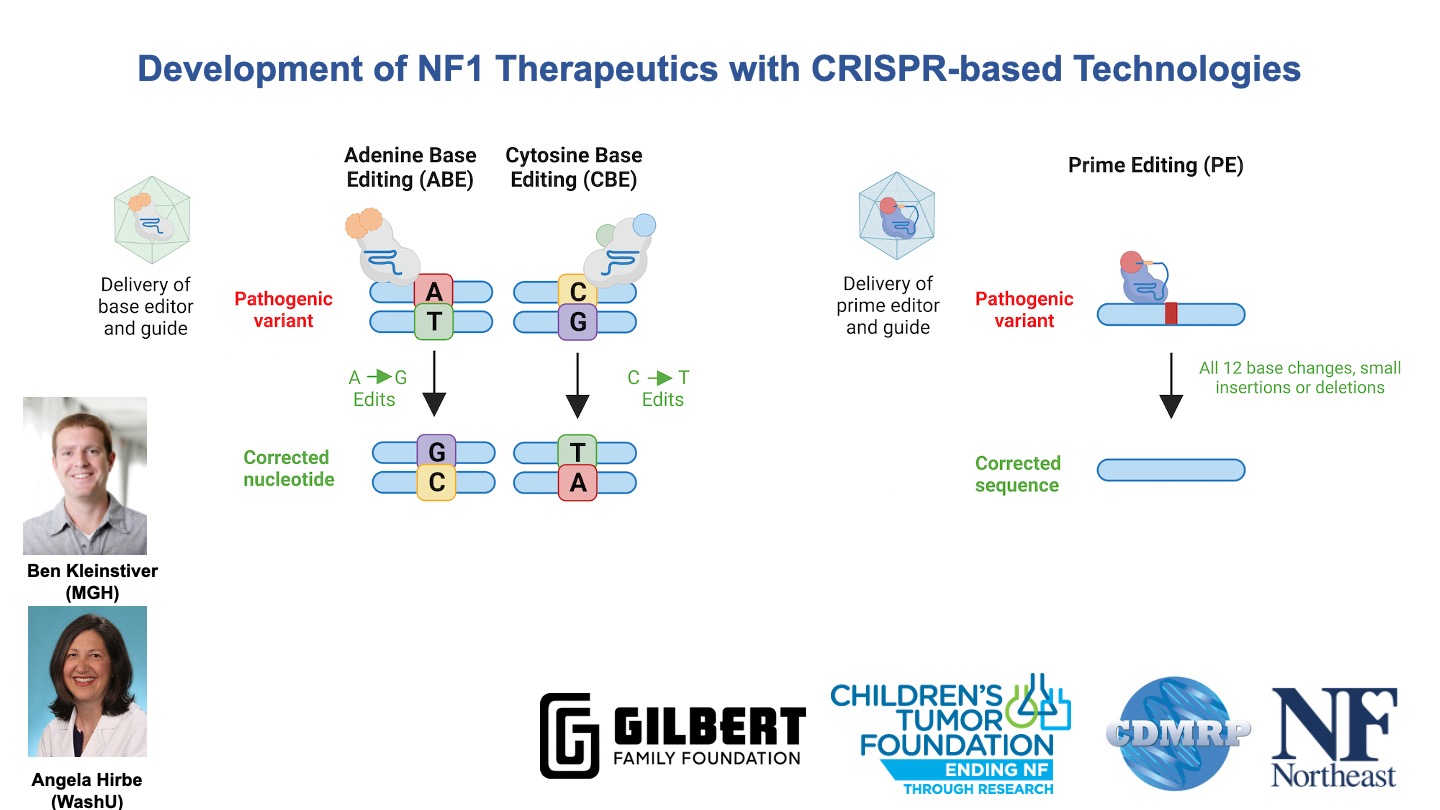
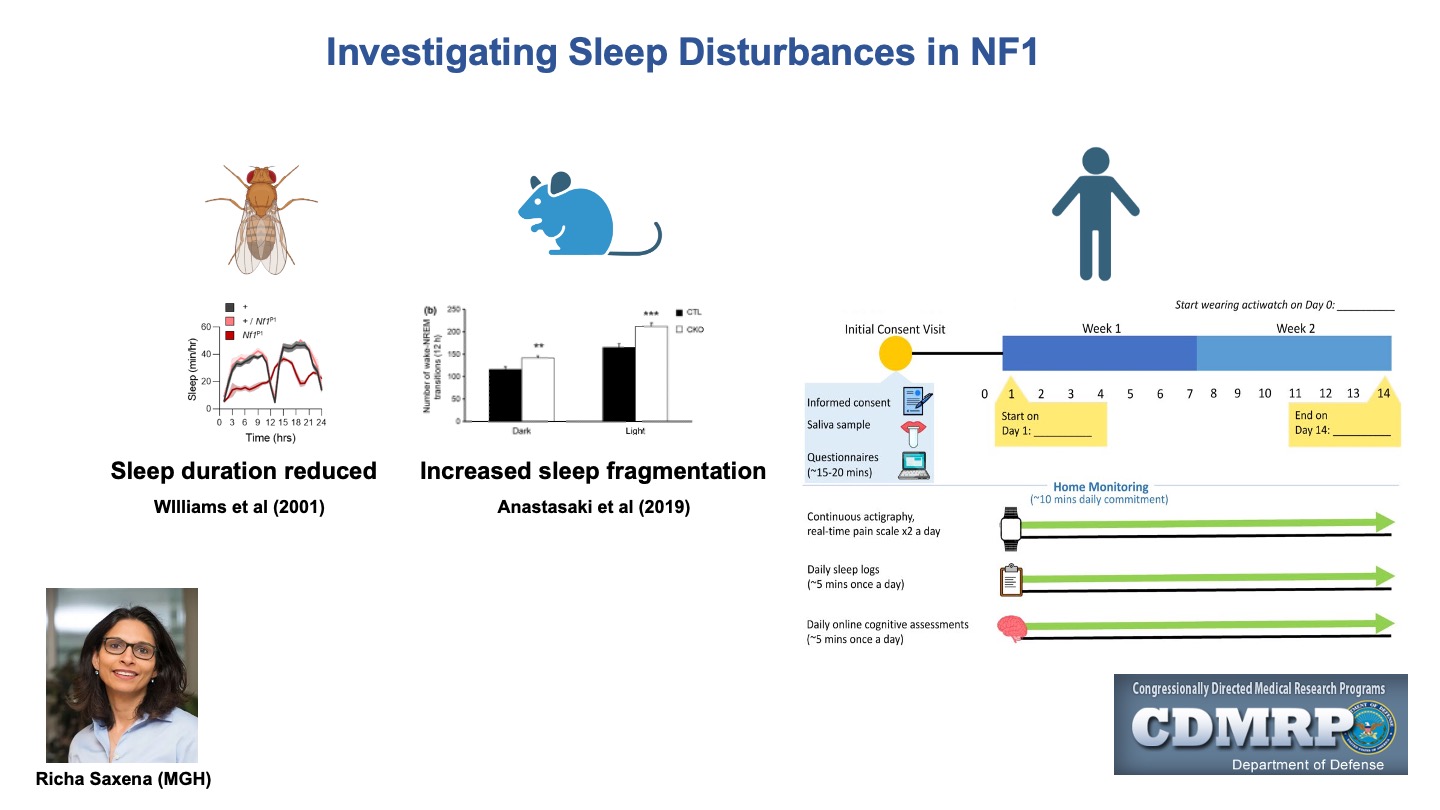
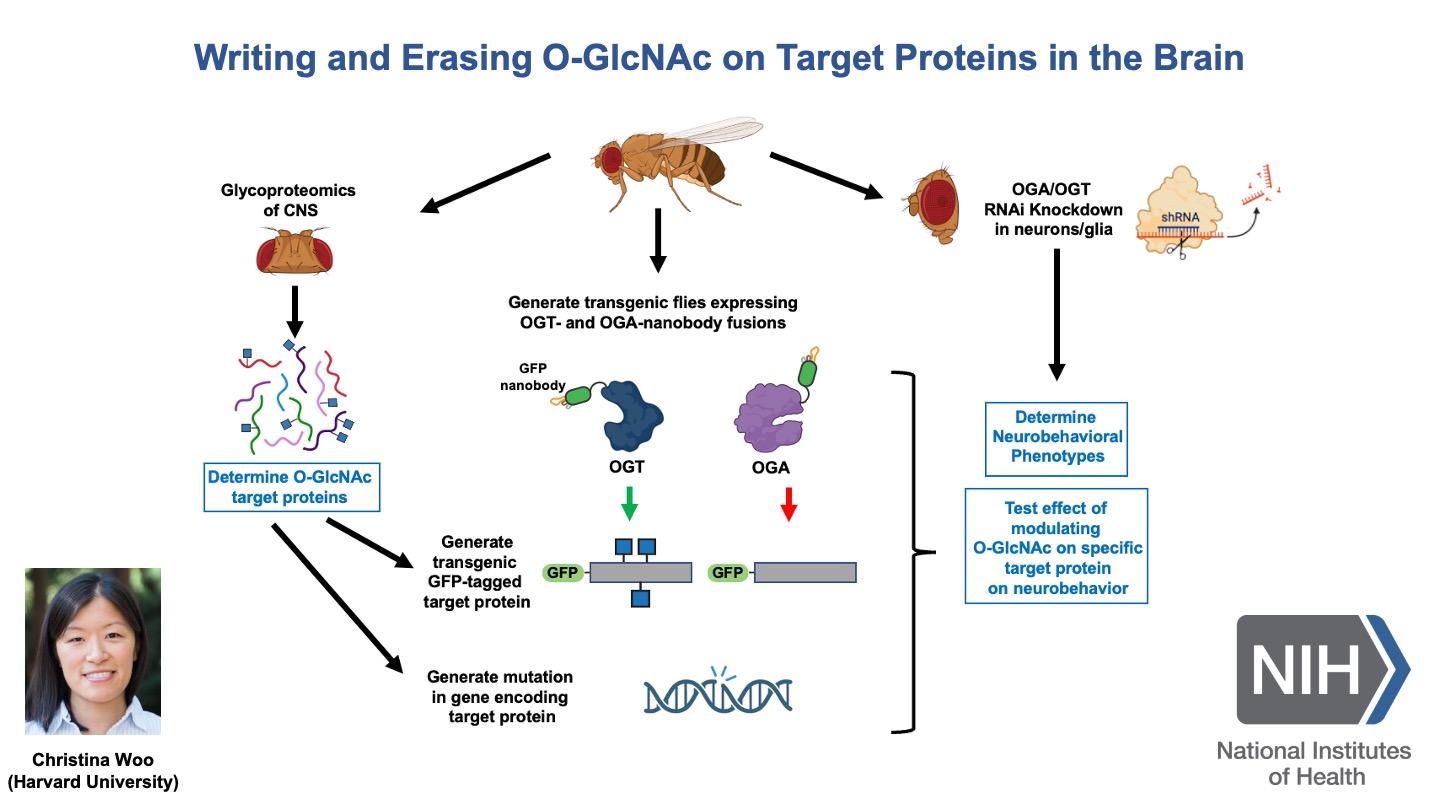
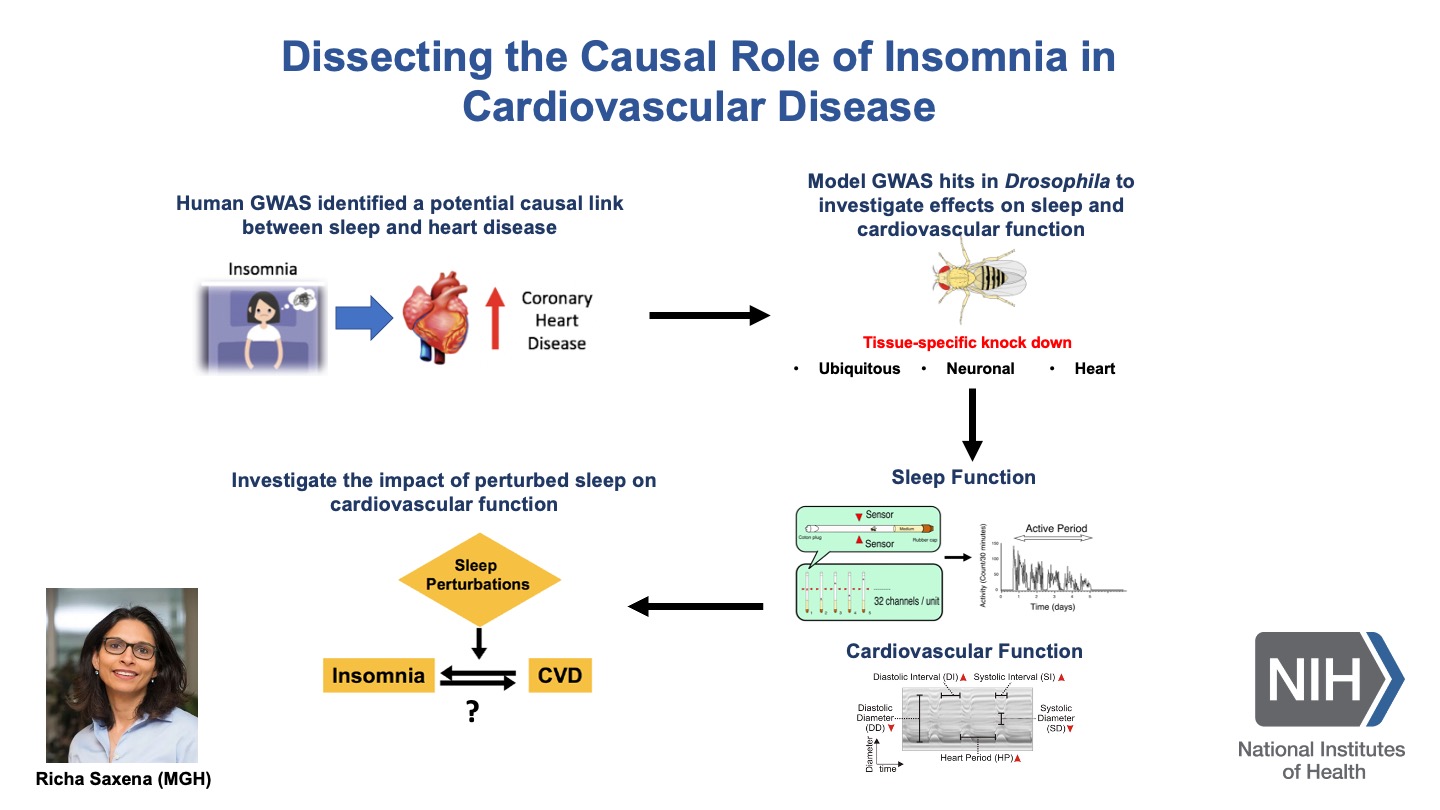
Some slides made using Biorender.com.